By Ann Reid
July 7, 2020
This post is part of a series exploring the lived experience of the 1918 influenza pandemic. Read the other posts in the series here.
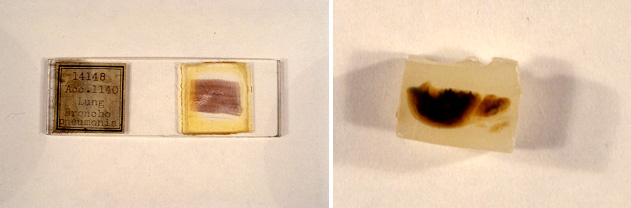
I had never heard of the 1918 influenza pandemic when my laboratory decided to try to decode the genetic sequence of the virus from archived samples at the Armed Forces Institute of Pathology (AFIP). The year was 1995, and I was in my mid-thirties. None of my contemporaries had ever heard of the pandemic either. So what made this a compelling scientific question? Despite its obscurity (the only book we could find about it, by Alfred Crosby, was entitled America’s Forgotten Pandemic), the pandemic raised important questions: why was the mortality rate so much higher than other pandemics? Why did it particularly target young adults? Where did the virus come from? Because scientists were unable to maintain viral cultures in the laboratory until more than a decade after the pandemic, no samples of the virus remained. Only by extracting the virus from contemporary samples would it be possible to decipher its genetic makeup. With the sequence of its genes in hand, perhaps its origin, virulence, and predilection for young adults could be explained.
The AFIP was uniquely placed to succeed in this, at the time, highly improbable venture. Its archive of conserved tissue samples from autopsies and biopsies dating back to the early twentieth century included over 70 samples from patients who had died in the pandemic. Furthermore, my colleagues and I had been working for several years to perfect the techniques needed to extract and sequence genetic material from archival samples. We were not optimistic about succeeding, but we knew that if we succeeded we would be making an important contribution to science.
Despite our ignorance of the pandemic, when I mentioned this project to friends, neighbors, and relatives in my parents’ generation—born in the 1920s or earlier—every one of them had a story to tell.
My next door neighbor, Frank, was born in 1917. In 1918, his mother passed away from the flu. He was raised by an aunt. My neighbor on the other side, Jack—at the time in his 60s—was an exception to the “born after 1930, never heard of the pandemic” rule. He had a story from his great-uncle, who had been a soldier in the U.S. Army, stationed in France in the fall of 1918.
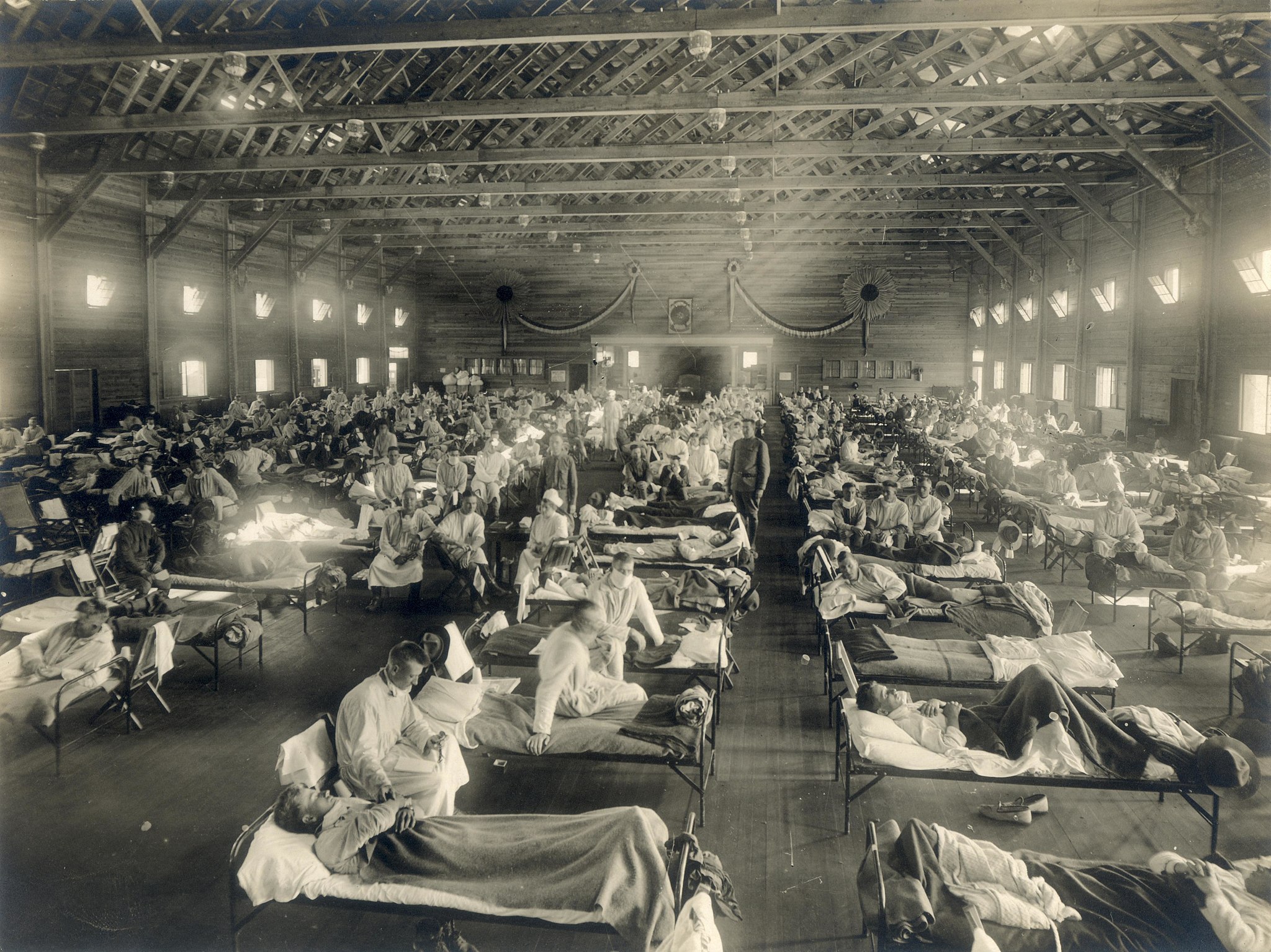
Jack’s great-uncle fell ill with the flu along with most of his fellow soldiers. They were cared for in a huge open ward—like those in the photos that have now become so familiar—meaning that they witnessed the often-fatal course of the disease in their friends. As Jack’s great-uncle told the story, he noticed that in the evening, nurses would sometimes give a few squares of chocolate to a patient. Invariably that patient was dead by morning. One evening, the nurse brought chocolate to Jack’s great-uncle, who refused it. He forever after ascribed his own survival to his decision not to eat the chocolate.
By 1995, there were relatively few people around who had been adults at the time of the pandemic. But most people who had been children in the 1920s and 1930s had heard of the pandemic and known of someone who had died—a parent, a local doctor or minister, a neighbor’s son who’d served in the Army or Navy. It seemed that even though almost everyone had personal knowledge of someone who had died, the pandemic was not talked about as a hugely significant event. Perhaps in a lifetime that included the Great Depression, the terrifying annual arrival of polio each summer, the Dust Bowl, and World War II, parents’ stories of a great flu outbreak just didn’t register as a big deal. And so those stories were not passed on to the next generation. By the 1970s, the pandemic was, indeed, forgotten.
Forgotten perhaps, but still compelling. I quickly became intrigued after reading Crosby’s book. Delving further into the contemporary scientific literature, especially the remarkable 1927 literature review, Influenza, compiled by Edwin O. Jordan (later a founder of the American Society for Microbiology) only increased my fascination, not to mention my respect for the dedicated and creative scientists who worked so hard to figure out what had happened. Meeting individual after individual who had stories to tell humanized the pandemic.

After our first publication, based on sequences obtained from the AFIP cases, we were contacted by a retired pathologist, Dr. Johan Hultin, who had unsuccessfully tried to revive the virus in 1953 from the bodies of Iñupiat villagers who had been buried in a mass grave in the permafrost on the coast of the Seward peninsula. In the town of Brevig Mission, 72 of 80 adult inhabitants died of the flu in November 1918. In 1999, Hultin returned to the village and, with the permission of the village elders, opened the grave again. We conducted the rest of our sequencing work on samples he sent back. Later we found an account of the outbreak in that very village by Clara Fosso, a Lutheran missionary. It haunts me still that her firsthand account may well mention the very woman whose remains eventually came into our hands.
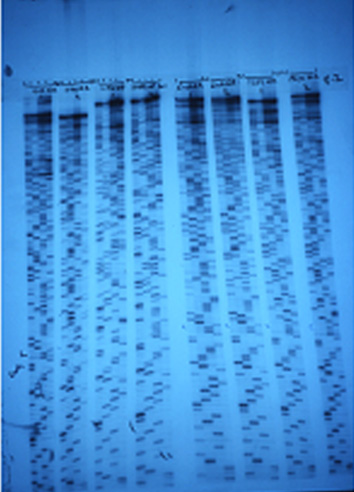
It took our team nearly a decade to determine the full genomic sequence of the virus. To our surprise and disappointment, the sequence provided limited clarity about the origin of the virus and no obvious explanation of its virulence, especially in such an unusual age group. Many scientists continue to work with the sequence, and clues have emerged about what made it so virulent. Now, as we live through another pandemic, scientists, physicians, and epidemiologists are again delving into contemporary accounts from 1918 to see what they can learn about the role of schools, masks, and social distancing in limiting pandemic spread. I suspect that, like me, they will find themselves relating to people long dead far more than they ever thought possible.
Ann Reid
Ann Reid is the executive director of the National Center for Science Education. For 15 years, she worked as a research biologist at the Armed Forces Institute of Pathology, where she was responsible for sequencing the 1918 flu virus. She has also served as a Senior Program Officer at the NRC’s Board on Life Sciences and as director of the American Academy of Microbiology.